Drug resistance threatens global malaria eradication efforts, necessitating innovative control strategies. This study examines how Mass Drug Administration (MDA) combined with antimalarial treatment can minimize resistance and improve malaria control.
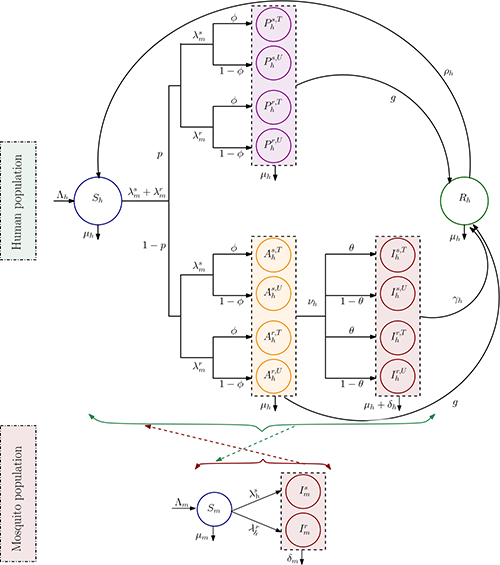
We developed a compartmental mathematical model to evaluate how resistance mutations affect transmission dynamics. The model tracks both treated and untreated populations among infected and recovered individuals, focusing on optimizing MDA and treatment strategies.
Global sensitivity analysis revealed critical factors influencing malaria dynamics: MDA coverage, treatment access across infection states, mutation probability from sensitive to resistant infections, and initial resistance prevalence. We extended the model to include optimal control strategies providing time-dependent interventions.
Results show that optimal strategies depend on mutation rates of sensitive infection to resistant infection. When mutation rates are low, the best approach combines antimalarial drugs with MDA, gradually reducing drug reliance over time for sustainable control. However, at higher mutation rates, strategies should prioritize broader MDA deployment while significantly limiting antimalarial use to prevent resistance development.
This research enhances understanding of how MDA and treatment strategies can effectively control malaria while managing resistance risks, providing valuable insights for optimizing malaria control programs in endemic regions. This study was conducted by Manuela Nimpa, PhD student under CRID’s Mathematical Modelling Unit.
Read more: https://www.aimspress.com/article/doi/10.3934/mbe.2025069